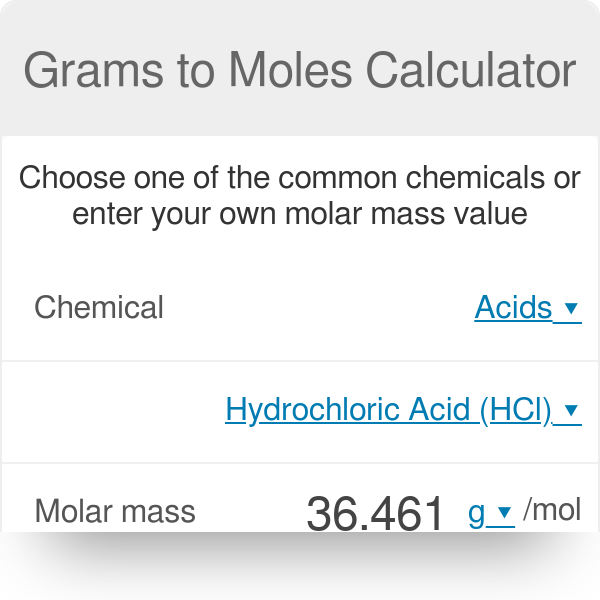
If the formula used in calculating molar mass is the molecular formula, the formula weight computed is the molecular weight. 
The formula weight is simply the weight in atomic mass units of all the atoms in a given formula. When calculating molecular weight of a chemical compound, it tells us how many grams are in one mole of that substance. 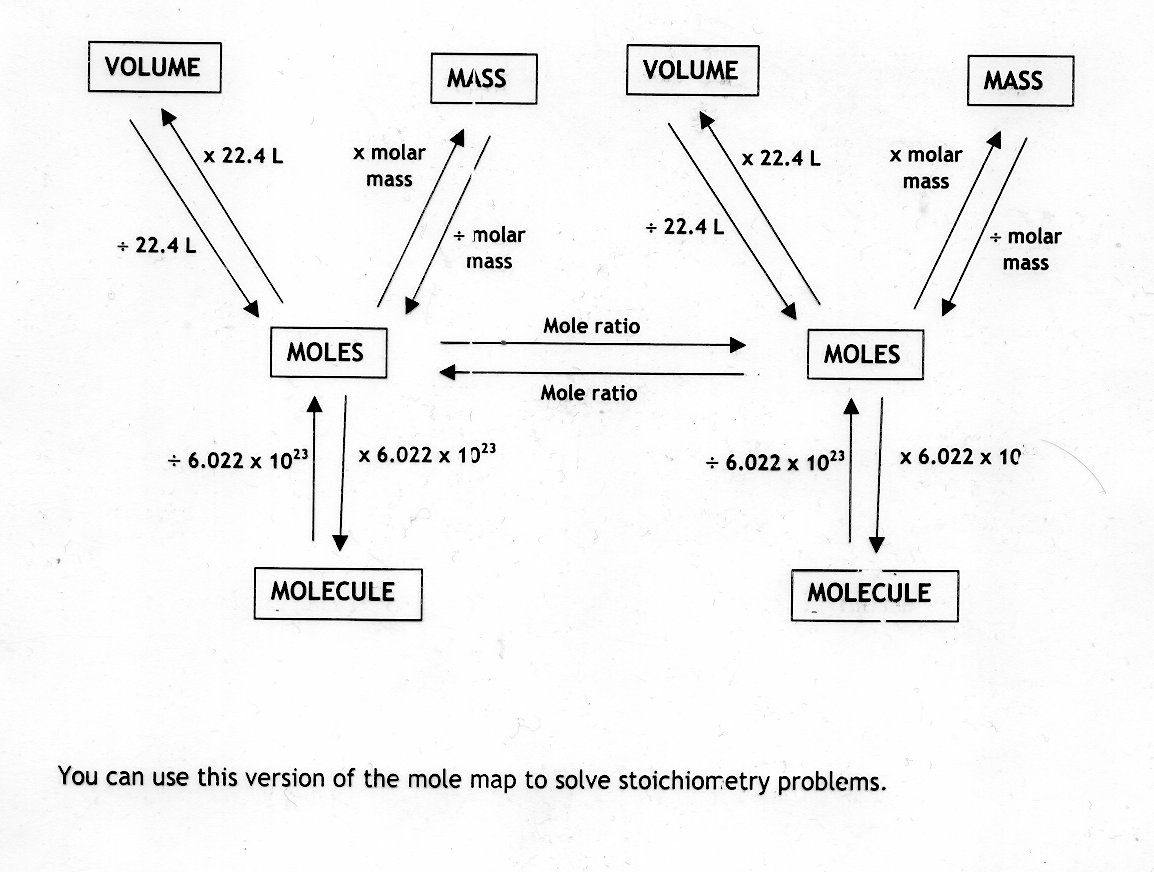
For bulk stoichiometric calculations, we are usually determining molar mass, which may also be called standard atomic weight or average atomic mass.įinding molar mass starts with units of grams per mole (g/mol). This is not the same as molecular mass, which is the mass of a single molecule of well-defined isotopes. This is how to calculate molar mass (average molecular weight), which is based on isotropically weighted averages. The atomic weights used on this site come from NIST, the National Institute of Standards and Technology. These relative weights computed from the chemical equation are sometimes called equation weights. In chemistry, the formula weight is a quantity computed by multiplying the atomic weight (in atomic mass units) of each element in a chemical formula by the number of atoms of that element present in the formula, then adding all of these products together.įormula weights are especially useful in determining the relative weights of reagents and products in a chemical reaction. Moles Carbon to grams, or enter other units to convert below: Enter two units to convert From: By understanding how to calculate moles using molecular weight, Avogadro’s number, or chemical equations, you will have a solid foundation for navigating the world of chemistry with ease and precision.You can do the reverse unit conversion from You can use these coefficients to determine the amount of reactants required or products formed for any given amount of substance participating in a reaction.Ĭalculating moles in chemistry is essential for accurately measuring and comparing quantities of substances involved in chemical reactions. In this balanced equation, 1 mole of methane reacts with 2 moles of oxygen to produce 1 mole of carbon dioxide and 2 moles of water. When working with balanced chemical equations, the coefficients (numbers in front of chemical Formulae) can be used to determine mole ratios between reactants and products.įor example, consider the combustion of methane: Moles = (Number of Particles) / (Avogadro’s Number)įor example, if your sample contains 3.012 × 10²³ atoms of carbon, you would divide that number by Avogadro’s number: 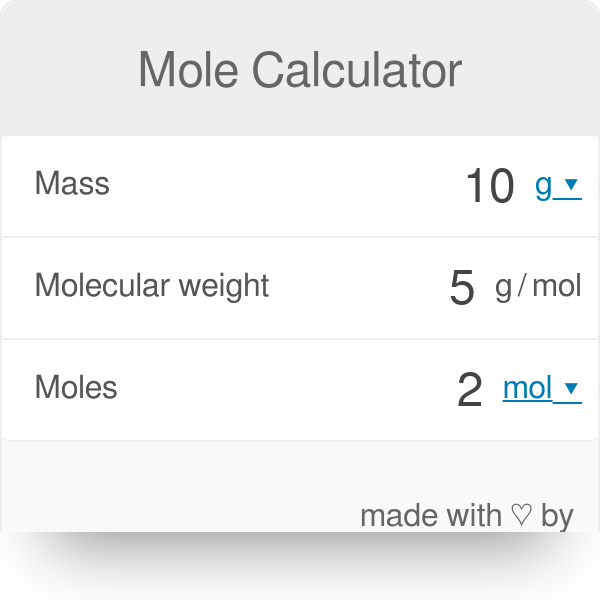
If you know the number of particles present in your sample and want to calculate moles, you can utilize Avogadro’s number (6.023 × 10²³): Now divide the mass by the molecular weight: Moles = (Mass of Substance) / (Molecular Weight)įor example, if you have 18 grams of water (H₂O), you would first find its molecular weight: H₂O = 2(1.01) + 16 = 18.02 g/mol. Molecular weight can be found on the periodic table or calculated by adding up the atomic masses of all atoms in a molecule. To calculate moles using molecular weight, you will need two pieces of information – the mass of the substance and its molecular weight (also called molar mass). By using moles, chemists can easily compare the amounts of various reactants and products involved in chemical reactions. The concept of the mole is crucial in chemistry because it provides a standardized method for expressing the amount of a substance in terms of its constituent particles. In this article, we will discuss the importance of moles in chemistry and outline the various methods to calculate moles for different chemical substances. A mole represents 6.023 × 10²³ particles, such as atoms, ions, or molecules. Moles are a fundamental unit of measurement in chemistry that is used to describe the amount of a substance.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
